We already have seed produced by the F1 plants created by the following cross:
|
|
|
- Timo Kokkonen
- 6 vuotta sitten
- Katselukertoja:
Transkriptio
1 Mapping the position of traits on a chromosome: Determining the genetic distance between a morphological trait and DNA Markers (Restriction Fragment Length Polymorphisms/RLFPs) using the model plant, Arabidopsis thaliana. You will use the genetic model plant, Arabidopsis thaliana, to conduct a linkage analysis of known Restriction Fragment Length Polymorphisms (RFLPs) and a mutation that causes a morphological defect called virescent. Arabidopsis is a small flowering plant with a short life cycle that is used as a model experimental system for studying plant biology. The A. thaliana genome, which is about 125 Mb, has been sequenced and annotated. There are many DNA markers scattered throughout A. thaliana s five chromosomes, including the three RFLPs on chromosome I that we will use in our analysis. Information on A. thaliana can be obtained at the web site, The vir2 mutation: The recessive mutation, vir2, causes leaves to be a lighter green than wild type and also has effects on growth (vir2/vir2 homozygous mutant plants are slower to germinate, grow more slowly, and are smaller in stature as compared to wild type). VIR2 is dominant and results in more rapid germination, dark leaf coloration, and larger plant stature. You will identify plants that are homozygous for vir2 by inspection, using VIR2/VIR2 wild type and vir2/vir2 mutant plants as controls for comparison. vir2 is known to be on chromosome I but its exact location is unknown. Your data will contribute to the BIO 304 database for mapping vir2. The RFLP Loci: You will use two RFLP loci in your attempt to map the location of vir2. RFLP #1 (R1) is located near the end of the left arm of chromosome 1, RFLP #2 (R2) is located near the end of the right arm of chromosome I. There are two alleles of each RFLP locus in the F2 population of plants you will study. The alleles of a given RFLP differ by the presence/absence of a single restriction enzyme site. To determine which RFLP alleles are present in a plant, you will amplify the locus by PCR and then digest the PCR product with the appropriate restriction enzyme and analyze the results by agarose gel electrophoresis. Restriction maps and the corresponding DNA sequences of both alleles of each locus are attached to this document. You should use this information to interpret the results of your analysis. Experimental Overview: Each student will select one vir2/vir2 F2 progeny plant from the cross series described below and analyze the plants DNA by PCR. You ll collect tissue samples from leaves using a Harris UniCore tissue sampler. You ll place a tissue sample from each plant in two tubes containing PCR reaction mixes. Each tube contains a reaction mix with primer pairs for amplifying one RFLP locus. You ll verify that the PCR reaction worked by gel electrophoresis and then digest a sample of the PCR product with the appropriate restriction enzyme and analyze the results by gel electrophoresis. You will use the information provided on the RFLP alleles to interpret your results and deduce the genotype of each plant. We already have seed produced by the F1 plants created by the following cross: P vir2 R1C R2C / vir2 R1C R2C VIR2 R1L R2L /VIR2 R1L R2L NOTE: The original parents include a vir2/vir2 mutant, which is in a genetic background known as Columbia. Columbia plants have the RFLP alleles R1C and R2C, where C stands for the Columbia allele. The other parent is a VIR2/VIR2 wild type in the Landsberg genetic background, which has the RFLP alleles R1L and R2L, where L stands for Landsberg. F1 F2 vir2 R1C R2C / VIR2 R1L R2L (these plants were allowed to self-pollinate to produce F2 seed) You will plant them on Day 1 of this lab.
2 Mapping a Morphological Trait Using DNA Markers (RFLPs): Lab 1 1. Sowing seeds from F1 plants (seeds are the F2 population) Today you will sow Arabidopsis seeds collected from F1 plants created by Biology personnel. The seeds are mixed with sand in a 1.5 ml microfuge tube. You to sprinkle the sand/seed mixture over the surface of soil in order to get an even distribution of F2 plants growing in your tray. Think of lightly salting food as you sprinkle the seed/sand mixture over the soil and avoid dropping a big clump of seed/sand in one spot. 2. Sowing seeds for controls The TAs have sown seed from the original parents (the Columbia and Landsberg ecotypes) and they will serve as controls for you to use as you try to identify VIR2+/ and vir2/vir2 among the F2. 3. Cold stratification to break seed dormancy The trays will be placed in a refrigerator for 1 week to simulate cold stratification, which occurs during the cold, moist conditions of winter in nature. Cold stratification helps break seed dormancy and, under laboratory conditions, promotes synchronous germination. 4. Coming up Five weeks from today you will observe the plants, score the vir2/vir2 morphological phenotype and take tissue samples for DNA analysis.
3 Your name and group #: Mapping a Morphological Trait Using DNA Markers (RFLPs): Report after Lab 1 1. Write the genotypes of P1, P2, and the F1. 2. Predict VIR2 and R1 genotypes of the gametes that were produced by the F1 plants. Indicate the proportion of each gamete genotype under each of the following assumptions: a) VIR2 and R1 assort independently b) VIF2 and R1 are 40 cm apart. c) VIR2 and R1 are 30 cm apart d) VIR2 and R1 are 20 cm apart e) VIR2 and R1 are 10 cm apart. Show your work below and record the genotype frequencies in Table 1. TABLE 1 UNLINKED 40cM apart 30cM apart 20cM apart 10cM apart vir2 - R1C vir2 - R1L VIR2 + R1C VIR2 + R1L
4 3. Predict VIR2 and R2 genotypes of the gametes that were produced by the F1 plants. Indicate the proportion of each gamete genotype under each of the following assumptions: a) VIR2 and R2 assort independently b) VIF2 and R2 are 40 cm apart. c) VIR2 and R2 are 30 cm apart d) VIR2 and R2 are 20 cm apart e) VIR2 and R2 are 10 cm apart. Show your work below and record your results in Table 2. TABLE 2 UNLINKED 40cM apart 30cM apart 20cM apart 10cM apart vir2 - R2C vir2 - R2L VIR2 + R2C VIR2 + R2L
5 RsaI (128) RsaI (90) RsaI (226) R1C PCR Product 941 bp 1 GTCCTATCTC TACGATGTGG ATGATATTAT TGCGGCGGAA GGTTCGATGA CAGGATAGAG ATGCTACACC TACTATAATA ACGCCGCCTT CCAAGCTACT 51 AGGGTAAACA AAAGCTGCCA CACACTGAAC AATTCCGGTA CGATTCTTTT TCCCATTTGT TTTCGACGGT GTGTGACTTG TTAAGGCCAT GCTAAGAAAA 101 AATTAACTCA TCATTTGCTT TCCACGTACG ATCCTCAAAA CTTACAATGA TTAATTGAGT AGTAAACGAA AGGTGCATGC TAGGAGTTTT GAATGTTACT 151 TTTAAATTTT AAGATAAAAG TTTTAAAATT TTTCTTTTTC TTTTCCATTT AAATTTAAAA TTCTATTTTC AAAATTTTAA AAAGAAAAAG AAAAGGTAAA 201 AAGGGTTAAT TGAAATAATT GAGGTACTGG TTCCCTTAAT TAAATGTGAT TTCCCAATTA ACTTTATTAA CTCCATGACC AAGGGAATTA ATTTACACTA 251 AGTATCAGTA GACTTTAAAA ATGTGAAATT AAAAGTGGGG TTGTGTGGGT TCATAGTCAT CTGAAATTTT TACACTTTAA TTTTCACCCC AACACACCCA 301 GAGAAATAAC TCGAAATGCT CGTGTCCTTT AATTATAGTG GGACCGTAAA CTCTTTATTG AGCTTTACGA GCACAGGAAA TTAATATCAC CCTGGCATTT 351 TATTTCTCAT TATATGCCTA CTTGGCATTA GAAATCATTT TTTTTTGACA ATAAAGAGTA ATATACGGAT GAACCGTAAT CTTTAGTAAA AAAAAACTGT 401 GCCATTATAA ATTAATTAAA AGGTGTTGAA TATCTTTCTT TGTCTTTTGT CGGTAATATT TAATTAATTT TCCACAACTT ATAGAAAGAA ACAGAAAACA 451 TACGGGGAAA ATAAATTATT TTCATAATAT TATTACTCGA CCATACAGCG ATGCCCCTTT TATTTAATAA AAGTATTATA ATAATGAGCT GGTATGTCGC 501 AAACTGGTCC AGGATACGAA TTATACAGCT AATTACAACA TTTTTGGATT TTTGACCAGG TCCTATGCTT AATATGTCGA TTAATGTTGT AAAAACCTAA 551 TTTGGGTTCA AAGTCCCTAC TTTGGCATTT TATATTGTAT ATTTTTTCCT AAACCCAAGT TTCAGGGATG AAACCGTAAA ATATAACATA TAAAAAAGGA 601 TAAAATCCTA TGTGTGTGTG CATGTGTATA TCTTTGTTAG ATAACACGTT ATTTTAGGAT ACACACACAC GTACACATAT AGAAACAATC TATTGTGCAA 651 AAGTTCGGTT CATAGAATAG GTAGGAAAAT AAAAGTAGTC TCTCGGAATT TTCAAGCCAA GTATCTTATC CATCCTTTTA TTTTCATCAG AGAGCCTTAA 701 AGTCGGAAAG ATAAAGTAAT TCAGATAAAC AAATGAATAA GTATATGCAA TCAGCCTTTC TATTTCATTA AGTCTATTTG TTTACTTATT CATATACGTT 751 GTGATATCAA GTGGGGTAAA ATTCTCCATC TTTACTTTTT GCCTATAAAA CACTATAGTT CACCCCATTT TAAGAGGTAG AAATGAAAAA CGGATATTTT 801 AATTTCACAC CCACTCGTAG AATATTTGGT TTTTACAGAA AGAGTATCTT TTAAAGTGTG GGTGAGCATC TTATAAACCA AAAATGTCTT TCTCATAGAA 851 CATCATTGCG GAAAATTAAA TACACCAATT TGCTATTTTA TCTTTATTAT GTAGTAACGC CTTTTAATTT ATGTGGTTAA ACGATAAAAT AGAAATAATA RsaI RsaI RsaI 901 AGTATTATAA TTATGATTAT GATTCTAATG CCTTATAACT T TCATAATATT AATACTAATA CTAAGATTAC GGAATATTGA A
6 RsaI (128) RsaI (90) R1L PCR Product 941 bp 1 GTCCTATCTC TACGATGTGG ATGATATTAT TGCGGCGGAA GGTTCGATGA CAGGATAGAG ATGCTACACC TACTATAATA ACGCCGCCTT CCAAGCTACT 51 AGGGTAAACA AAAGCTGCCA CACACTGAAC AATTCCGGTA CGATTCTTTT TCCCATTTGT TTTCGACGGT GTGTGACTTG TTAAGGCCAT GCTAAGAAAA 101 AATTAACTCA TCATTTGCTT TCCACGTACG ATCCTCAAAA CTTACAATGA TTAATTGAGT AGTAAACGAA AGGTGCATGC TAGGAGTTTT GAATGTTACT 151 TTTAAATTTT AAGATAAAAG TTTTAAAATT TTTCTTTTTC TTTTCCATTT AAATTTAAAA TTCTATTTTC AAAATTTTAA AAAGAAAAAG AAAAGGTAAA 201 AAGGGTTAAT TGAAATAATT GAGGTGCTGG TTCCCTTAAT TAAATGTGAT TTCCCAATTA ACTTTATTAA CTCCACGACC AAGGGAATTA ATTTACACTA 251 AGTATCAGTA GACTTTAAAA ATGTGAAATT AAAAGTGGGG TTGTGTGGGT TCATAGTCAT CTGAAATTTT TACACTTTAA TTTTCACCCC AACACACCCA 301 GAGAAATAAC TCGAAATGCT CGTGTCCTTT AATTATAGTG GGACCGTAAA CTCTTTATTG AGCTTTACGA GCACAGGAAA TTAATATCAC CCTGGCATTT 351 TATTTCTCAT TATATGCCTA CTTGGCATTA GAAATCATTT TTTTTTGACA ATAAAGAGTA ATATACGGAT GAACCGTAAT CTTTAGTAAA AAAAAACTGT 401 GCCATTATAA ATTAATTAAA AGGTGTTGAA TATCTTTCTT TGTCTTTTGT CGGTAATATT TAATTAATTT TCCACAACTT ATAGAAAGAA ACAGAAAACA 451 TACGGGGAAA ATAAATTATT TTCATAATAT TATTACTCGA CCATACAGCG ATGCCCCTTT TATTTAATAA AAGTATTATA ATAATGAGCT GGTATGTCGC 501 AAACTGGTCC AGGATACGAA TTATACAGCT AATTACAACA TTTTTGGATT TTTGACCAGG TCCTATGCTT AATATGTCGA TTAATGTTGT AAAAACCTAA 551 TTTGGGTTCA AAGTCCCTAC TTTGGCATTT TATATTGTAT ATTTTTTCCT AAACCCAAGT TTCAGGGATG AAACCGTAAA ATATAACATA TAAAAAAGGA 601 TAAAATCCTA TGTGTGTGTG CATGTGTATA TCTTTGTTAG ATAACACGTT ATTTTAGGAT ACACACACAC GTACACATAT AGAAACAATC TATTGTGCAA 651 AAGTTCGGTT CATAGAATAG GTAGGAAAAT AAAAGTAGTC TCTCGGAATT TTCAAGCCAA GTATCTTATC CATCCTTTTA TTTTCATCAG AGAGCCTTAA 701 AGTCGGAAAG ATAAAGTAAT TCAGATAAAC AAATGAATAA GTATATGCAA TCAGCCTTTC TATTTCATTA AGTCTATTTG TTTACTTATT CATATACGTT 751 GTGATATCAA GTGGGGTAAA ATTCTCCATC TTTACTTTTT GCCTATAAAA CACTATAGTT CACCCCATTT TAAGAGGTAG AAATGAAAAA CGGATATTTT 801 AATTTCACAC CCACTCGTAG AATATTTGGT TTTTACAGAA AGAGTATCTT TTAAAGTGTG GGTGAGCATC TTATAAACCA AAAATGTCTT TCTCATAGAA 851 CATCATTGCG GAAAATTAAA TACACCAATT TGCTATTTTA TCTTTATTAT GTAGTAACGC CTTTTAATTT ATGTGGTTAA ACGATAAAAT AGAAATAATA RsaI RsaI 901 AGTATTATAA TTATGATTAT GATTCTAATG CCTTATAACT T TCATAATATT AATACTAATA CTAAGATTAC GGAATATTGA A
7 1 GCGTGACCAT CAAGACTAAT TAACTAAGAC CACATTTTAA AAAAACTATT CGCACTGGTA GTTCTGATTA ATTGATTCTG GTGTAAAATT TTTTTGATAA 51 AATAATTACT ACAATTTGTA ATTAAAAAGA TCAACGAGAA ATGCCACGTG TTATTAATGA TGTTAAACAT TAATTTTTCT AGTTGCTCTT TACGGTGCAC 101 GACGAATACT AGCAACGCCA AGTGGAAAGA GCGTTCGAGA GAACAAGGCA CTGCTTATGA TCGTTGCGGT TCACCTTTCT CGCAAGCTCT CTTGTTCCGT 151 AAACCAAATA CGCCCCTAGT ATTCTACAGA TGTCGACTGG ATAATTACAA TTTGGTTTAT GCGGGGATCA TAAGATGTCT ACAGCTGACC TATTAATGTT 201 AAGATTTCAA TAAACAGTAC TAATTAATTT CTAGTGGTGA GTTTTTGTAA TTCTAAAGTT ATTTGTCATG ATTAATTAAA GATCACCACT CAAAAACATT 251 ATATCTACTT CTTCCAATTA CCAGCTGCTA TATAAATCCC CTTCTCTGTT TATAGATGAA GAAGGTTAAT GGTCGACGAT ATATTTAGGG GAAGAGACAA 301 TCTCTTTTCT TACATCACAA TCACACAAAA CTAACAAAAG ATCAAAAGCA AGAGAAAAGA ATGTAGTGTT AGTGTGTTTT GATTGTTTTC TAGTTTTCGT 351 AGTTCTTCAC TGTTGATAAT GTCTACCACC GGACAGATTA TTCGATGCAA TCAAGAAGTG ACAACTATTA CAGATGGTGG CCTGTCTAAT AAGCTACGTT 401 AGGTTTTCTT TTTATTCTGT CTTTTTCCAA ATATTTATTG ATCGGTTACA TCCAAAAGAA AAATAAGACA GAAAAAGGTT TATAAATAAC TAGCCAATGT 451 TTTCTGTTGA GGTTTTTGTT ATGAATCCAC AATTTCTATG TTGAATTAAC AAAGACAACT CCAAAAACAA TACTTAGGTG TTAAAGATAC AACTTAATTG 501 AAAACCTGTG TCGTTTTTTT GTGGTGGTTG CAGCTGCTGT GGCATGGGAA TTTTGGACAC AGCAAAAAAA CACCACCAAC GTCGACGACA CCGTACCCTT 551 GCCGGAAAGC CACTGGTGAT CGAGGAAGTG GAGGTTGCTC CACCGCAGAA CGGCCTTTCG GTGACCACTA GCTCCTTCAC CTCCAACGAG GTGGCGTCTT 601 ACACGAAGTT CGTATCAAGA TTCTCTTCAC TTCTCTCTGT CACACCGATG TGTGCTTCAA GCATAGTTCT AAGAGAAGTG AAGAGAGACA GTGTGGCTAC 651 TTTACTTCTG GGAAGCTAAG GTAGAGTAAT CAATTTATTA CACTCCAAAT AAATGAAGAC CCTTCGATTC CATCTCATTA GTTAAATAAT GTGAGGTTTA 701 TCATAATCAA GTTCTAATTT TTTTAGAATT CTAATTTTTT ATCTAAAAAA AGTATTAGTT CAAGATTAAA AAAATCTTAA GATTAAAAAA TAGATTTTTT 751 ATTCAACCTT TTTGATTCCA CAGGGACAAA CACCGTTGTT TCCACGTATC TAAGTTGGAA AAACTAAGGT GTCCCTGTTT GTGGCAACAA AGGTGCATAG 801 TTCGGCCATG AAGCTGGAGG GTAATAGAAA CACTAATCTT CTTTGCTTCG AAGCCGGTAC TTCGACCTCC CATTATCTTT GTGATTAGAA GAAACGAAGC 851 TTTTGGATAT TTTTAAGGTT TTAGAGATTC AAGGTCGTTT TTTTTGTTGT AAAACCTATA AAAATTCCAA AATCTCTAAG TTCCAGCAAA AAAAACAACA 901 TGTGTAGGAT TGTTGAGAGT GTTGGAGAAG GAGTGACTGA TCTTCAGCCA ACACATCCTA ACAACTCTCA CAACCTCTTC CTCACTGACT AGAAGTCGGT 951 GGAGATCATG TGTTGCCGAT CTTTACCGGA GAATGTGGGG AGTGTCGTCA CCTCTAGTAC ACAACGGCTA GAAATGGCCT CTTACACCCC TCACAGCAGT 1001 TTGCCACTCG GAGGAATCAA ACATGTGTGA TCTTCTCAGG ATCAACACCG AACGGTGAGC CTCCTTAGTT TGTACACACT AGAAGAGTCC TAGTTGTGGC 1051 AGCGAGGAGG GATGATTCAC GATGGTGAAT CAAGATTCTC CATTAATGGC TCGCTCCTCC CTACTAAGTG CTACCACTTA GTTCTAAGAG GTAATTACCG 1101 AAACCAATTT ACCATTTCCT TGGGACTTCC ACGTTCAGTG AGTACACAGT TTTGGTTAAA TGGTAAAGGA ACCCTGAAGG TGCAAGTCAC TCATGTGTCA 1151 GGTTCACTCT GGTCAGGTTG CTAAGATCAA TCCGGATGCT CCTCTTGACA CCAAGTGAGA CCAGTCCAAC GATTCTAGTT AGGCCTACGA GGAGAACTGT 1201 AGGTCTGTAT TGTCAGTTGT GGTTTGTCTA CTGGGTTAGG AGCAACTTTG TCCAGACATA ACAGTCAACA CCAAACAGAT GACCCAATCC TCGTTGAAAC 1251 AATGTGGCTA AACCCAAGAA AGGTCAAAGT GTTGCCATTT TT TTACACCGAT TTGGGTTCTT TCCAGTTTCA CAACGGTAAA AA 1292 bp R2C PCR Product
8 1 GCGTGACCAT CAAGACTAAT TAACTAAGAC CACATTTTAA AAAAACTATT CGCACTGGTA GTTCTGATTA ATTGATTCTG GTGTAAAATT TTTTTGATAA 51 AATAATTACT ACAATTTGTA ATTAAAAAGA TCAACGAGAA ATGCCACGTG TTATTAATGA TGTTAAACAT TAATTTTTCT AGTTGCTCTT TACGGTGCAC 101 GACGAATACT AGCAACGCCA AGTGGAAAGA GCGTTCGAGA GAACAAGGCA CTGCTTATGA TCGTTGCGGT TCACCTTTCT CGCAAGCTCT CTTGTTCCGT 151 AAACCAAATA CGCCCCTAGT ATTCTACAGA TGTCGACTGG ATAATTACAA TTTGGTTTAT GCGGGGATCA TAAGATGTCT ACAGCTGACC TATTAATGTT 201 AAGATTTCAA TAAACAGTAC TAATTAATTT CTAGTGGTGA GTTTTTGTAA TTCTAAAGTT ATTTGTCATG ATTAATTAAA GATCACCACT CAAAAACATT 251 ATATCTACTT CTTCCAATTA CCAGCTGCTA TATAAATCCC CTTCTCTGTT TATAGATGAA GAAGGTTAAT GGTCGACGAT ATATTTAGGG GAAGAGACAA 301 TCTCTTTTCT TACATCACAA TCACACAAAA CTAACAAAAG ATCAAAAGCA AGAGAAAAGA ATGTAGTGTT AGTGTGTTTT GATTGTTTTC TAGTTTTCGT 351 AGTTCTTCAC TGTTGATAAT GTCTACCACC GGACAGATTA TTCGATGCAA TCAAGAAGTG ACAACTATTA CAGATGGTGG CCTGTCTAAT AAGCTACGTT 401 AGGTTTTCTT TTTATTCTGT CTTTTTCCAA ATATTTATTG ATCGGTTACA TCCAAAAGAA AAATAAGACA GAAAAAGGTT TATAAATAAC TAGCCAATGT 451 TTTCTGTTGA GGTTTTTGTT ATGAATCCAC AATTTCTATG TTGAATTAAC AAAGACAACT CCAAAAACAA TACTTAGGTG TTAAAGATAC AACTTAATTG 501 AAAACCTGTG TCGTTTTTTT GTGGTGGTTG CAGCTGCTGT GGCATGGGAA TTTTGGACAC AGCAAAAAAA CACCACCAAC GTCGACGACA CCGTACCCTT 551 GCCGGAAAGC CACTGGTGAT CGAGGAAGTG GAGGTTGCTC CACCGCAGAA CGGCCTTTCG GTGACCACTA GCTCCTTCAC CTCCAACGAG GTGGCGTCTT 601 ACACGAAGTT CGTATCAAGA TTCTCTTCAC TTCTCTCTGT CACACCGATG TGTGCTTCAA GCATAGTTCT AAGAGAAGTG AAGAGAGACA GTGTGGCTAC 651 TTTACTTCTG GGAAGCTAAG GTAGAGTAAT CAATTTATTA CACTCCAAAT AAATGAAGAC CCTTCGATTC CATCTCATTA GTTAAATAAT GTGAGGTTTA 701 TCATAATCAA GTTCTAATTT TTTTAGAATT CTAATTTTTT ATCTAAAAAA AGTATTAGTT CAAGATTAAA AAAATCTTAA GATTAAAAAA TAGATTTTTT 751 ATTCAACCTT TTTGATTCCA CAGGGACAAA CACCGTTGTT TCCACGTATC TAAGTTGGAA AAACTAAGGT GTCCCTGTTT GTGGCAACAA AGGTGCATAG 801 TTCGGCCATG AAGCTGGAGG GTAATAGAAA CACTAATCTT CTTTGCTTCG AAGCCGGTAC TTCGACCTCC CATTATCTTT GTGATTAGAA GAAACGAAGC 851 TTTTGGATAT TTTTAAGGTT TTAGAGATTC AAGGTCGTTT TTTTTGTTGT AAAACCTATA AAAATTCCAA AATCTCTAAG TTCCAGCAAA AAAAACAACA 901 TGTGTAGGAT TGTTGAGAGT GTTGGAGAAG GAGTGACTGA TCTTCAGCCA ACACATCCTA ACAACTCTCA CAACCTCTTC CTCACTGACT AGAAGTCGGT 951 GGAGATCATG TGTTGCCGAT CTTTACCGGA GAATGTGGGG AGTGTCGTCA CCTCTAGTAC ACAACGGCTA GAAATGGCCT CTTACACCCC TCACAGCAGT 1001 TTGCCACTCG GAGGAATCAA ACATGTGTGA TCTTCTCAGG ATCAACACCG AACGGTGAGC CTCCTTAGTT TGTACACACT AGAAGAGTCC TAGTTGTGGC 1051 AGCGAGGAGG GATGATTCAC GATGGTGAAT CTAGATTCTC CATTAATGGC TCGCTCCTCC CTACTAAGTG CTACCACTTA GATCTAAGAG GTAATTACCG 1101 AAACCAATTT ACCATTTCCT TGGGACTTCC ACGTTCAGTG AGTACACAGT TTTGGTTAAA TGGTAAAGGA ACCCTGAAGG TGCAAGTCAC TCATGTGTCA 1151 GGTTCACTCT GGTCAGGTTG CTAAGATCAA TCCGGATGCT CCTCTTGACA CCAAGTGAGA CCAGTCCAAC GATTCTAGTT AGGCCTACGA GGAGAACTGT 1201 AGGTCTGTAT TGTCAGTTGT GGTTTGTCTA CTGGGTTAGG AGCAACTTTG TCCAGACATA ACAGTCAACA CCAAACAGAT GACCCAATCC TCGTTGAAAC 1251 AATGTGGCTA AACCCAAGAA AGGTCAAAGT GTTGCCATTT TT TTACACCGAT TTGGGTTCTT TCCAGTTTCA CAACGGTAAA AA XbaI 1292 bp R2L PCR Product XbaI (1081)
9 Mapping a Morphological Trait Using DNA Markers (RFLPs): Lab 2 Today you will observe the phenotype of your F2 plants, count the number of wild type and vir2 mutant plants, and set up PCR reactions to amplify three RFLP loci from tissue sample of two vir2 mutant plants. A. On your table you will have: Two PCR reaction tubes R1 (blue) and R2 (orange/red) 1 ml of bleach 1ml of sterile distilled water A Harris 0.5 mm UniCore hole punch (don t touch it yet!) A cutting pad Plants PCR contents: 20 ul reaction total volume 10 ul 2X buffer 1 ul forward primer 1 ul reverse primer 7.6 ul dh 2 O 0.4 ul enzyme B. Count the number of wild type and mutant F2 plants in the tray. We ll take the average of the class data. Wild type: Mutant: Record your results in the table on the Smart Board and record class data. C. Obtain leaf tissue samples for PCR: EACH STUDENT ANALZES ONE PLANT You will take tissue plugs from a leaf on each plant you analyze and add it to a PCR reaction mixture provided to you by the TAs. You will do this by using a 0.5 mm Harris hole punch to obtain leaf tissue cores, using the technique illustrated by the instructor. PLEASE NOTE: 1. Do not stab yourself with the needle of the hole punch. You should analyze plant DNA, not your own. 2. Do not break the needle of the hole punch. Very little force/pressure is needed to push the needle through the leaf, so don t over exert yourself and break the hole punch. 3. Rinse the hole punch needle in water between punches of the same leaf a. Rinse the hole punch by submerging the needle in water and pressing the button ten times. 4. Wash and rinse the hole punch when you are done with one leaf and before you work on another leaf. a. Wash the hole punch needle in bleach by submerging the needle in bleach and pressing the button three times. b. After the bleach wash, rinse the hole punch by submerging the needle in water and pressing the button ten times.
10 Detailed Protocol for Setting up PCR Reactions: 1. Each student should select one (supposed) vir2/vir2 plant for analysis. 2. Take one leaf from one plant; place it on the cutting pad. Extract a tissue plug from the leaf using the Harris hole punch as demonstrated by the instructor and eject the plug into the blue R1 tube. Make sure the leaf plug is visible in the tube using the dissecting scope and then close the tube tightly. Label it R1 and your assigned student number (1 through 30). 3. Rinse the hole punch in water. Extract a second plug from the same leaf and eject it into the orange/red R2 tube. Make sure the leaf plug is visible in the tube using the dissecting scope and then close the tube tightly. Label it R2 and your assigned student number. 4. Wash the hole punch in bleach and then rinse in water. ALL BLEACH MUST BE RINSED OFF! 5. Let another student at your table use the punch and pad for their plant leaf. 6. When all students are done, wash the hole punch in bleach and rinse it in water and leave it on your bench for a TA to collect. 7. Inform the TAs that you are done and they will collect the PCR reactions, place them in a PCR machine and run the following PCR protocol: 1 Cycle of 98 o C for 5 minutes 40 Cycles of 98 o C for 5 seconds 58 o C for 5 seconds 72 o C for 30 seconds 1 Cycle of 72 o C for 1 minute You will analyze the PCR reactions in the next lab period.
11 Your name and group #: Mapping a Morphological Trait Using DNA Markers (RFLPs): Report after Lab 2 1. The seeds you plated a few weeks back were supposed to be F2 from the series of crosses presented at the beginning of this lab and which you wrote up for the lab 1. There is some doubt among the BIO 304 instructors and TA whether this is true because the mutant phenotype has not been obvious among potential F2 populations in semesters past. State the class observations and perform a chi-square analysis to determine whether the observed proportion of vir2/vir2 plants is consistent with the plants being F2. 2. The attached sequence pages show the R1 and R2 genetic loci that will be amplified by the PCR reactions you set up today. The PCR reaction includes a step to allow primers to anneal to the templates ( annealing step ), which is conducted at a temperature that is determined by the melting temperature of the primers in each reaction. The primers for the R1 locus correspond to the first 23 bp of R1 sequence and the last 27 bp of the sequence. The primers for the R2 locus correspond to the first 20 bp and last 20 bp of R2. Write the primer sequences and calculate the T m of these primers using the following equation: #G+#C-16.4 N o T m( C)= ( )
12 3. Use the sequence information for R1 and R2 to determine the expected size of each PCR product. Also indicate the size of restriction fragments that will be produced for the C and L alleles of each locus. Enter your information in the table below. Locus R1C R1L R2C R2L Size of Undigested PCR Product Size of Fragments After Restriction Digests 4. Since it was difficult to identify vir2/ vir2 mutants, it is possible that someone messed up and the seeds planted at the beginning of this lab were not F2. There are no other A. thaliana seed sources in the department, therefore the possible alternatives are P1 (Columbia ecotype), P2 (Landsberg ecotype), or the F1 hybrid. The class has performed PCR analysis of two RFLP loci of many plants. Predict the results of the RFLP analysis if the plants are P1, P2 or F1 using the information from step 3. Predicted RLFP data if the plants are P1: R1 locus: R2 locus: Predicted RFLP data if the plants are P2: R1 locus R2 locus Predicted RFLP data if the plants are F1: R1 locus R2 locus
13 Mapping a Morphological Trait Using DNA Markers (RFLPs): Lab 3 Today you will run samples of your PCR reactions from the previous lab on a gel and set up restriction digests of each PCR reactions. You will analyze the restriction digests during the next lab period. A. On your bench you will have: PCR reactions you set up in the previous lab period a tube with DNA molecular weight markers a tube with 6X gel loading buffer a 1.6% agarose gel in the gel tank Two tubes containing 15 ul of RsaI Reaction Mix (BLUE TUBES) Two tubes containing 15 ul of XbaI Reaction Mix (ORANGE TUBES) B. Set up restriction digests of a 5 ul sample of each PCR reaction by pipetting 5 ul of the PCR reaction into the appropriate Reaction Mix tube: 5 ul of the one student s R1 PCR reaction into one Blue Tube. Label it with the student # 5 ul of that same student s R2 PCR reaction into one Orange Tube. Label it with the student # 5 ul of the another student s R1 PCR reaction into one Blue Tube. Label it with the student # 5 ul of that same student s R2 PCR reaction into one Orange Tube. Label it with the student # NOTE: continue until each student has set up one digest for each of their PCR reaction. Incubate the reactions at 37 degrees for 30 minutes, and then prepare the reaction for gel electrophoresis by adding 5ul of 6X gel loading buffer. C. Load sample of your restriction digests on your gel as follows: 10 ul molecular weight marker in lane 1 20 ul of one student s Blue Tube in lane 2 20 ul of a second student s Blue Tube in lane 3 20 ul of a third student s Blue Tube in lane 4 20 ul of the first student s Orange Tube in lane 5 20 ul of the second student s Orange Tube in lane 6 20 ul of the third student s Orange Tube in lane 7 NOTE: The other two students on the bench should use the second gel to run markers and their digests using the same pattern as above. Run the gel at 90 volts for about 30 minutes. The TAs will photograph the gel and give you a copy of the photo. D. Analyze your results: Analyze your results using the information from your write up from plant lab 2. Plant 1: vir2 vir2, R1 R1, R2 R2
14 Record your results on the white board and record your data and the class data in Table 4 on the next page. You will use this information to determine whether there is evidence for linkage between the vir2 locus and each RFLP locus. Class results: Student # vir2 vir2 R1 R1 R2 R2 1 vir2 vir2 2 vir2 vir2 3 vir2 vir2 4 vir2 vir2 5 vir2 vir2 6 vir2 vir2 7 vir2 vir2 8 vir2 vir2 9 vir2 vir2 10 vir2 vir2 11 vir2 vir2 12 vir2 vir2 13 vir2 vir2 14 vir2 vir2 15 vir2 vir2 16 vir2 vir2 17 vir2 vir2 18 vir2 vir2 19 vir2 vir2 20 vir2 vir2 21 vir2 vir2 22 vir2 vir2 23 vir2 vir2 24 vir2 vir2 25 vir2 vir2 26 vir2 vir2 27 vir2 vir2 28 vir2 vir2 29 vir2 vir2 30 vir2 vir2 31 vir2 vir2 32 vir2 vir2 33 vir2 vir2 34 vir2 vir2 35 vir2 vir2
15 Your name and group #: Mapping a Morphological Trait Using DNA Markers (RFLPs): Report after Lab 4 1. Based on the class data, what is the percent recombination for vir2 and the R1 locus? 2. Perform a chi-square test to determine whether the evidence for linkage of vir2 and R1 is statistically significant. Show your work below. 3. Based on the class data, what is the percent recombination for vir2 and the R1 locus? 4. Perform a chi-square test to determine whether the evidence for linkage of vir2 and R1 is statistically significant. Show your work below. 5. Draw a map of chromosome I showing the locations of the R1, R2, and VIR2 loci.
1. SIT. The handler and dog stop with the dog sitting at heel. When the dog is sitting, the handler cues the dog to heel forward.
 START START SIT 1. SIT. The handler and dog stop with the dog sitting at heel. When the dog is sitting, the handler cues the dog to heel forward. This is a static exercise. SIT STAND 2. SIT STAND. The
START START SIT 1. SIT. The handler and dog stop with the dog sitting at heel. When the dog is sitting, the handler cues the dog to heel forward. This is a static exercise. SIT STAND 2. SIT STAND. The
tgg agg Supplementary Figure S1.
 ttaggatattcggtgaggtgatatgtctctgtttggaaatgtctccgccattaactcaag tggaaagtgtatagtaatgaatctttcaagcacacagatcacttcaaaagactgtttcaa catcacctcaggacaaaaagatgtactctcatttggatgctgtgatgccatgggtcacag attgcaattcccaagtgcccgttcttttacaccaaaatcaaagaagaatatctccccttt
ttaggatattcggtgaggtgatatgtctctgtttggaaatgtctccgccattaactcaag tggaaagtgtatagtaatgaatctttcaagcacacagatcacttcaaaagactgtttcaa catcacctcaggacaaaaagatgtactctcatttggatgctgtgatgccatgggtcacag attgcaattcccaagtgcccgttcttttacaccaaaatcaaagaagaatatctccccttt
On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31)
 On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31) Juha Kahkonen Click here if your download doesn"t start automatically On instrument costs
On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31) Juha Kahkonen Click here if your download doesn"t start automatically On instrument costs
Capacity Utilization
 Capacity Utilization Tim Schöneberg 28th November Agenda Introduction Fixed and variable input ressources Technical capacity utilization Price based capacity utilization measure Long run and short run
Capacity Utilization Tim Schöneberg 28th November Agenda Introduction Fixed and variable input ressources Technical capacity utilization Price based capacity utilization measure Long run and short run
Choose Finland-Helsinki Valitse Finland-Helsinki
 Write down the Temporary Application ID. If you do not manage to complete the form you can continue where you stopped with this ID no. Muista Temporary Application ID. Jos et onnistu täyttää lomake loppuun
Write down the Temporary Application ID. If you do not manage to complete the form you can continue where you stopped with this ID no. Muista Temporary Application ID. Jos et onnistu täyttää lomake loppuun
Efficiency change over time
 Efficiency change over time Heikki Tikanmäki Optimointiopin seminaari 14.11.2007 Contents Introduction (11.1) Window analysis (11.2) Example, application, analysis Malmquist index (11.3) Dealing with panel
Efficiency change over time Heikki Tikanmäki Optimointiopin seminaari 14.11.2007 Contents Introduction (11.1) Window analysis (11.2) Example, application, analysis Malmquist index (11.3) Dealing with panel
FinFamily PostgreSQL installation ( ) FinFamily PostgreSQL
 FinFamily PostgreSQL 1 Sisällys / Contents FinFamily PostgreSQL... 1 1. Asenna PostgreSQL tietokanta / Install PostgreSQL database... 3 1.1. PostgreSQL tietokannasta / About the PostgreSQL database...
FinFamily PostgreSQL 1 Sisällys / Contents FinFamily PostgreSQL... 1 1. Asenna PostgreSQL tietokanta / Install PostgreSQL database... 3 1.1. PostgreSQL tietokannasta / About the PostgreSQL database...
Network to Get Work. Tehtäviä opiskelijoille Assignments for students. www.laurea.fi
 Network to Get Work Tehtäviä opiskelijoille Assignments for students www.laurea.fi Ohje henkilöstölle Instructions for Staff Seuraavassa on esitetty joukko tehtäviä, joista voit valita opiskelijaryhmällesi
Network to Get Work Tehtäviä opiskelijoille Assignments for students www.laurea.fi Ohje henkilöstölle Instructions for Staff Seuraavassa on esitetty joukko tehtäviä, joista voit valita opiskelijaryhmällesi
On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31)
 On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31) Juha Kahkonen Click here if your download doesn"t start automatically On instrument costs
On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31) Juha Kahkonen Click here if your download doesn"t start automatically On instrument costs
Information on preparing Presentation
 Information on preparing Presentation Seminar on big data management Lecturer: Spring 2017 20.1.2017 1 Agenda Hints and tips on giving a good presentation Watch two videos and discussion 22.1.2017 2 Goals
Information on preparing Presentation Seminar on big data management Lecturer: Spring 2017 20.1.2017 1 Agenda Hints and tips on giving a good presentation Watch two videos and discussion 22.1.2017 2 Goals
The CCR Model and Production Correspondence
 The CCR Model and Production Correspondence Tim Schöneberg The 19th of September Agenda Introduction Definitions Production Possiblity Set CCR Model and the Dual Problem Input excesses and output shortfalls
The CCR Model and Production Correspondence Tim Schöneberg The 19th of September Agenda Introduction Definitions Production Possiblity Set CCR Model and the Dual Problem Input excesses and output shortfalls
anna minun kertoa let me tell you
 anna minun kertoa let me tell you anna minun kertoa I OSA 1. Anna minun kertoa sinulle mitä oli. Tiedän että osaan. Kykenen siihen. Teen nyt niin. Minulla on oikeus. Sanani voivat olla puutteellisia mutta
anna minun kertoa let me tell you anna minun kertoa I OSA 1. Anna minun kertoa sinulle mitä oli. Tiedän että osaan. Kykenen siihen. Teen nyt niin. Minulla on oikeus. Sanani voivat olla puutteellisia mutta
Results on the new polydrug use questions in the Finnish TDI data
 Results on the new polydrug use questions in the Finnish TDI data Multi-drug use, polydrug use and problematic polydrug use Martta Forsell, Finnish Focal Point 28/09/2015 Martta Forsell 1 28/09/2015 Esityksen
Results on the new polydrug use questions in the Finnish TDI data Multi-drug use, polydrug use and problematic polydrug use Martta Forsell, Finnish Focal Point 28/09/2015 Martta Forsell 1 28/09/2015 Esityksen
Other approaches to restrict multipliers
 Other approaches to restrict multipliers Heikki Tikanmäki Optimointiopin seminaari 10.10.2007 Contents Short revision (6.2) Another Assurance Region Model (6.3) Cone-Ratio Method (6.4) An Application of
Other approaches to restrict multipliers Heikki Tikanmäki Optimointiopin seminaari 10.10.2007 Contents Short revision (6.2) Another Assurance Region Model (6.3) Cone-Ratio Method (6.4) An Application of
National Building Code of Finland, Part D1, Building Water Supply and Sewerage Systems, Regulations and guidelines 2007
 National Building Code of Finland, Part D1, Building Water Supply and Sewerage Systems, Regulations and guidelines 2007 Chapter 2.4 Jukka Räisä 1 WATER PIPES PLACEMENT 2.4.1 Regulation Water pipe and its
National Building Code of Finland, Part D1, Building Water Supply and Sewerage Systems, Regulations and guidelines 2007 Chapter 2.4 Jukka Räisä 1 WATER PIPES PLACEMENT 2.4.1 Regulation Water pipe and its
1. Liikkuvat määreet
 1. Liikkuvat määreet Väitelauseen perussanajärjestys: SPOTPA (subj. + pred. + obj. + tapa + paikka + aika) Suora sanajärjestys = subjekti on ennen predikaattia tekijä tekeminen Alasääntö 1: Liikkuvat määreet
1. Liikkuvat määreet Väitelauseen perussanajärjestys: SPOTPA (subj. + pred. + obj. + tapa + paikka + aika) Suora sanajärjestys = subjekti on ennen predikaattia tekijä tekeminen Alasääntö 1: Liikkuvat määreet
16. Allocation Models
 16. Allocation Models Juha Saloheimo 17.1.27 S steemianalsin Optimointiopin seminaari - Sks 27 Content Introduction Overall Efficienc with common prices and costs Cost Efficienc S steemianalsin Revenue
16. Allocation Models Juha Saloheimo 17.1.27 S steemianalsin Optimointiopin seminaari - Sks 27 Content Introduction Overall Efficienc with common prices and costs Cost Efficienc S steemianalsin Revenue
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.9.269
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.9.269
On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31)
 On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31) Juha Kahkonen Click here if your download doesn"t start automatically On instrument costs
On instrument costs in decentralized macroeconomic decision making (Helsingin Kauppakorkeakoulun julkaisuja ; D-31) Juha Kahkonen Click here if your download doesn"t start automatically On instrument costs
Lab SBS3.FARM_Hyper-V - Navigating a SharePoint site
 Lab SBS3.FARM_Hyper-V - Navigating a SharePoint site Note! Before starting download and install a fresh version of OfficeProfessionalPlus_x64_en-us. The instructions are in the beginning of the exercise.
Lab SBS3.FARM_Hyper-V - Navigating a SharePoint site Note! Before starting download and install a fresh version of OfficeProfessionalPlus_x64_en-us. The instructions are in the beginning of the exercise.
MALE ADULT FIBROBLAST LINE (82-6hTERT)
 Double-stranded methylation patterns of a 104-bp L1 promoter in DNAs from male and female fibroblasts, male leukocytes and female lymphoblastoid cells using hairpin-bisulfite PCR. Fifteen L1 sequences
Double-stranded methylation patterns of a 104-bp L1 promoter in DNAs from male and female fibroblasts, male leukocytes and female lymphoblastoid cells using hairpin-bisulfite PCR. Fifteen L1 sequences
Metsälamminkankaan tuulivoimapuiston osayleiskaava
 VAALAN KUNTA TUULISAIMAA OY Metsälamminkankaan tuulivoimapuiston osayleiskaava Liite 3. Varjostusmallinnus FCG SUUNNITTELU JA TEKNIIKKA OY 12.5.2015 P25370 SHADOW - Main Result Assumptions for shadow calculations
VAALAN KUNTA TUULISAIMAA OY Metsälamminkankaan tuulivoimapuiston osayleiskaava Liite 3. Varjostusmallinnus FCG SUUNNITTELU JA TEKNIIKKA OY 12.5.2015 P25370 SHADOW - Main Result Assumptions for shadow calculations
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
Plasmid Name: pmm290. Aliases: none known. Length: bp. Constructed by: Mike Moser/Cristina Swanson. Last updated: 17 August 2009
 Plasmid Name: pmm290 Aliases: none known Length: 11707 bp Constructed by: Mike Moser/Cristina Swanson Last updated: 17 August 2009 Description and application: This is a mammalian expression vector for
Plasmid Name: pmm290 Aliases: none known Length: 11707 bp Constructed by: Mike Moser/Cristina Swanson Last updated: 17 August 2009 Description and application: This is a mammalian expression vector for
TM ETRS-TM35FIN-ETRS89 WTG
 VE1 SHADOW - Main Result Calculation: 8 x Nordex N131 x HH145m Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please
VE1 SHADOW - Main Result Calculation: 8 x Nordex N131 x HH145m Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
( ( OX2 Perkkiö. Rakennuskanta. Varjostus. 9 x N131 x HH145
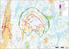 OX2 9 x N131 x HH145 Rakennuskanta Asuinrakennus Lomarakennus Liike- tai julkinen rakennus Teollinen rakennus Kirkko tai kirkollinen rak. Muu rakennus Allas Varjostus 1 h/a 8 h/a 20 h/a 0 0,5 1 1,5 2 km
OX2 9 x N131 x HH145 Rakennuskanta Asuinrakennus Lomarakennus Liike- tai julkinen rakennus Teollinen rakennus Kirkko tai kirkollinen rak. Muu rakennus Allas Varjostus 1 h/a 8 h/a 20 h/a 0 0,5 1 1,5 2 km
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
FETAL FIBROBLASTS, PASSAGE 10
 Double-stranded methylation patterns of a 104-bp L1 promoter in DNAs from fetal fibroblast passages 10, 14, 17, and 22 using barcoded hairpinbisulfite PCR. Fifteen L1 sequences were analyzed for passages
Double-stranded methylation patterns of a 104-bp L1 promoter in DNAs from fetal fibroblast passages 10, 14, 17, and 22 using barcoded hairpinbisulfite PCR. Fifteen L1 sequences were analyzed for passages
Tynnyrivaara, OX2 Tuulivoimahanke. ( Layout 9 x N131 x HH145. Rakennukset Asuinrakennus Lomarakennus 9 x N131 x HH145 Varjostus 1 h/a 8 h/a 20 h/a
 , Tuulivoimahanke Layout 9 x N131 x HH145 Rakennukset Asuinrakennus Lomarakennus 9 x N131 x HH145 Varjostus 1 h/a 8 h/a 20 h/a 0 0,5 1 1,5 km 2 SHADOW - Main Result Assumptions for shadow calculations
, Tuulivoimahanke Layout 9 x N131 x HH145 Rakennukset Asuinrakennus Lomarakennus 9 x N131 x HH145 Varjostus 1 h/a 8 h/a 20 h/a 0 0,5 1 1,5 km 2 SHADOW - Main Result Assumptions for shadow calculations
Experimental Identification and Computational Characterization of a Novel. Extracellular Metalloproteinase Produced by Clostridium sordellii
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 207 Supplementary Information Experimental Identification and Computational Characterization of
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 207 Supplementary Information Experimental Identification and Computational Characterization of
Uusi Ajatus Löytyy Luonnosta 4 (käsikirja) (Finnish Edition)
 Uusi Ajatus Löytyy Luonnosta 4 (käsikirja) (Finnish Edition) Esko Jalkanen Click here if your download doesn"t start automatically Uusi Ajatus Löytyy Luonnosta 4 (käsikirja) (Finnish Edition) Esko Jalkanen
Uusi Ajatus Löytyy Luonnosta 4 (käsikirja) (Finnish Edition) Esko Jalkanen Click here if your download doesn"t start automatically Uusi Ajatus Löytyy Luonnosta 4 (käsikirja) (Finnish Edition) Esko Jalkanen
make and make and make ThinkMath 2017
 Adding quantities Lukumäärienup yhdistäminen. Laske yhteensä?. Countkuinka howmonta manypalloja ballson there are altogether. and ja make and make and ja make on and ja make ThinkMath 7 on ja on on Vaihdannaisuus
Adding quantities Lukumäärienup yhdistäminen. Laske yhteensä?. Countkuinka howmonta manypalloja ballson there are altogether. and ja make and make and ja make on and ja make ThinkMath 7 on ja on on Vaihdannaisuus
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
Methods S1. Sequences relevant to the constructed strains, Related to Figures 1-6.
 Methods S1. Sequences relevant to the constructed strains, Related to Figures 1-6. A. Promoter Sequences Gal4 binding sites are highlighted in the color referenced in Figure 1A when possible. Site 1: red,
Methods S1. Sequences relevant to the constructed strains, Related to Figures 1-6. A. Promoter Sequences Gal4 binding sites are highlighted in the color referenced in Figure 1A when possible. Site 1: red,
FIS IMATRAN KYLPYLÄHIIHDOT Team captains meeting
 FIS IMATRAN KYLPYLÄHIIHDOT 8.-9.12.2018 Team captains meeting 8.12.2018 Agenda 1 Opening of the meeting 2 Presence 3 Organizer s personell 4 Jury 5 Weather forecast 6 Composition of competitors startlists
FIS IMATRAN KYLPYLÄHIIHDOT 8.-9.12.2018 Team captains meeting 8.12.2018 Agenda 1 Opening of the meeting 2 Presence 3 Organizer s personell 4 Jury 5 Weather forecast 6 Composition of competitors startlists
Statistical design. Tuomas Selander
 Statistical design Tuomas Selander 28.8.2014 Introduction Biostatistician Work area KYS-erva KYS, Jyväskylä, Joensuu, Mikkeli, Savonlinna Work tasks Statistical methods, selection and quiding Data analysis
Statistical design Tuomas Selander 28.8.2014 Introduction Biostatistician Work area KYS-erva KYS, Jyväskylä, Joensuu, Mikkeli, Savonlinna Work tasks Statistical methods, selection and quiding Data analysis
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
Bounds on non-surjective cellular automata
 Bounds on non-surjective cellular automata Jarkko Kari Pascal Vanier Thomas Zeume University of Turku LIF Marseille Universität Hannover 27 august 2009 J. Kari, P. Vanier, T. Zeume (UTU) Bounds on non-surjective
Bounds on non-surjective cellular automata Jarkko Kari Pascal Vanier Thomas Zeume University of Turku LIF Marseille Universität Hannover 27 august 2009 J. Kari, P. Vanier, T. Zeume (UTU) Bounds on non-surjective
WindPRO version joulu 2012 Printed/Page :47 / 1. SHADOW - Main Result
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
KONEISTUSKOKOONPANON TEKEMINEN NX10-YMPÄRISTÖSSÄ
 KONEISTUSKOKOONPANON TEKEMINEN NX10-YMPÄRISTÖSSÄ https://community.plm.automation.siemens.com/t5/tech-tips- Knowledge-Base-NX/How-to-simulate-any-G-code-file-in-NX- CAM/ta-p/3340 Koneistusympäristön määrittely
KONEISTUSKOKOONPANON TEKEMINEN NX10-YMPÄRISTÖSSÄ https://community.plm.automation.siemens.com/t5/tech-tips- Knowledge-Base-NX/How-to-simulate-any-G-code-file-in-NX- CAM/ta-p/3340 Koneistusympäristön määrittely
VAASAN YLIOPISTO Humanististen tieteiden kandidaatin tutkinto / Filosofian maisterin tutkinto
 VAASAN YLIOPISTO Humanististen tieteiden kandidaatin tutkinto / Filosofian maisterin tutkinto Tämän viestinnän, nykysuomen ja englannin kandidaattiohjelman valintakokeen avulla Arvioidaan viestintävalmiuksia,
VAASAN YLIOPISTO Humanististen tieteiden kandidaatin tutkinto / Filosofian maisterin tutkinto Tämän viestinnän, nykysuomen ja englannin kandidaattiohjelman valintakokeen avulla Arvioidaan viestintävalmiuksia,
,0 Yes ,0 120, ,8
 SHADOW - Main Result Calculation: Alue 2 ( x 9 x HH120) TuuliSaimaa kaavaluonnos Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered
SHADOW - Main Result Calculation: Alue 2 ( x 9 x HH120) TuuliSaimaa kaavaluonnos Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table 5.11.2013 16:44 / 1 Minimum
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table 5.11.2013 16:44 / 1 Minimum
Gap-filling methods for CH 4 data
 Gap-filling methods for CH 4 data Sigrid Dengel University of Helsinki Outline - Ecosystems known for CH 4 emissions; - Why is gap-filling of CH 4 data not as easy and straight forward as CO 2 ; - Gap-filling
Gap-filling methods for CH 4 data Sigrid Dengel University of Helsinki Outline - Ecosystems known for CH 4 emissions; - Why is gap-filling of CH 4 data not as easy and straight forward as CO 2 ; - Gap-filling
WindPRO version joulu 2012 Printed/Page :42 / 1. SHADOW - Main Result
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table 13.6.2013 19:42 / 1 Minimum
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table 13.6.2013 19:42 / 1 Minimum
You can check above like this: Start->Control Panel->Programs->find if Microsoft Lync or Microsoft Lync Attendeed is listed
 Online Meeting Guest Online Meeting for Guest Participant Lync Attendee Installation Online kokous vierailevalle osallistujalle Lync Attendee Asennus www.ruukki.com Overview Before you can join to Ruukki
Online Meeting Guest Online Meeting for Guest Participant Lync Attendee Installation Online kokous vierailevalle osallistujalle Lync Attendee Asennus www.ruukki.com Overview Before you can join to Ruukki
Research plan for masters thesis in forest sciences. The PELLETime 2009 Symposium Mervi Juntunen
 Modelling tree and stand characteristics and estimating biomass removals and harvesting costs of lodgepole pine (Pinus contorta) plantations in Iceland Research plan for masters thesis in forest sciences
Modelling tree and stand characteristics and estimating biomass removals and harvesting costs of lodgepole pine (Pinus contorta) plantations in Iceland Research plan for masters thesis in forest sciences
The Viking Battle - Part Version: Finnish
 The Viking Battle - Part 1 015 Version: Finnish Tehtävä 1 Olkoon kokonaisluku, ja olkoon A n joukko A n = { n k k Z, 0 k < n}. Selvitä suurin kokonaisluku M n, jota ei voi kirjoittaa yhden tai useamman
The Viking Battle - Part 1 015 Version: Finnish Tehtävä 1 Olkoon kokonaisluku, ja olkoon A n joukko A n = { n k k Z, 0 k < n}. Selvitä suurin kokonaisluku M n, jota ei voi kirjoittaa yhden tai useamman
( ,5 1 1,5 2 km
 Tuulivoimala Rakennukset Asuinrakennus Liikerak. tai Julkinen rak. Lomarakennus Teollinen rakennus Kirkollinen rakennus Varjostus "real case" h/a 1 h/a 8 h/a 20 h/a 4 5 3 1 2 6 7 8 9 10 0 0,5 1 1,5 2 km
Tuulivoimala Rakennukset Asuinrakennus Liikerak. tai Julkinen rak. Lomarakennus Teollinen rakennus Kirkollinen rakennus Varjostus "real case" h/a 1 h/a 8 h/a 20 h/a 4 5 3 1 2 6 7 8 9 10 0 0,5 1 1,5 2 km
LYTH-CONS CONSISTENCY TRANSMITTER
 LYTH-CONS CONSISTENCY TRANSMITTER LYTH-INSTRUMENT OY has generate new consistency transmitter with blade-system to meet high technical requirements in Pulp&Paper industries. Insurmountable advantages are
LYTH-CONS CONSISTENCY TRANSMITTER LYTH-INSTRUMENT OY has generate new consistency transmitter with blade-system to meet high technical requirements in Pulp&Paper industries. Insurmountable advantages are
Curriculum. Gym card
 A new school year Curriculum Fast Track Final Grading Gym card TET A new school year Work Ethic Detention Own work Organisation and independence Wilma TMU Support Services Well-Being CURRICULUM FAST TRACK
A new school year Curriculum Fast Track Final Grading Gym card TET A new school year Work Ethic Detention Own work Organisation and independence Wilma TMU Support Services Well-Being CURRICULUM FAST TRACK
MEETING PEOPLE COMMUNICATIVE QUESTIONS
 Tiistilän koulu English Grades 7-9 Heikki Raevaara MEETING PEOPLE COMMUNICATIVE QUESTIONS Meeting People Hello! Hi! Good morning! Good afternoon! How do you do? Nice to meet you. / Pleased to meet you.
Tiistilän koulu English Grades 7-9 Heikki Raevaara MEETING PEOPLE COMMUNICATIVE QUESTIONS Meeting People Hello! Hi! Good morning! Good afternoon! How do you do? Nice to meet you. / Pleased to meet you.
Ajettavat luokat: SM: S1 (25 aika-ajon nopeinta)
 SUPERMOTO SM 2013 OULU Lisämääräys ja ohje Oulun Moottorikerho ry ja Oulun Formula K-125ry toivottaa SuperMoto kuljettajat osallistumaan SuperMoto SM 2013 Oulu osakilpailuun. Kilpailu ajetaan karting radalla
SUPERMOTO SM 2013 OULU Lisämääräys ja ohje Oulun Moottorikerho ry ja Oulun Formula K-125ry toivottaa SuperMoto kuljettajat osallistumaan SuperMoto SM 2013 Oulu osakilpailuun. Kilpailu ajetaan karting radalla
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table 22.12.2014 11:33 / 1 Minimum
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table 22.12.2014 11:33 / 1 Minimum
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Calculation: N117 x 9 x HH141 Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG
SHADOW - Main Result Calculation: N117 x 9 x HH141 Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG
Returns to Scale II. S ysteemianalyysin. Laboratorio. Esitelmä 8 Timo Salminen. Teknillinen korkeakoulu
 Returns to Scale II Contents Most Productive Scale Size Further Considerations Relaxation of the Convexity Condition Useful Reminder Theorem 5.5 A DMU found to be efficient with a CCR model will also be
Returns to Scale II Contents Most Productive Scale Size Further Considerations Relaxation of the Convexity Condition Useful Reminder Theorem 5.5 A DMU found to be efficient with a CCR model will also be
Opiskelijoiden ajatuksia koulun alkuun liittyen / students thoughts about the beginning of their studies at KSYK
 Opiskelijoiden ajatuksia koulun alkuun liittyen / students thoughts about the beginning of their studies at KSYK Helppoa/mukavaa/palkitsevaa - easy/nice/rewarding - uudet ystävät/ new friends - koulun
Opiskelijoiden ajatuksia koulun alkuun liittyen / students thoughts about the beginning of their studies at KSYK Helppoa/mukavaa/palkitsevaa - easy/nice/rewarding - uudet ystävät/ new friends - koulun
TM ETRS-TM35FIN-ETRS89 WTG
 SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
SHADOW - Main Result Assumptions for shadow calculations Maximum distance for influence Calculate only when more than 20 % of sun is covered by the blade Please look in WTG table WindPRO version 2.8.579
Travel Getting Around
 - Location Olen eksyksissä. Not knowing where you are Voisitko näyttää kartalta missä sen on? Asking for a specific location on a map Mistä täällä on? Asking for a specific...wc?...pankki / rahanvaihtopiste?...hotelli?...huoltoasema?...sairaala?...apteekki?...tavaratalo?...ruokakauppa?...bussipysäkki?
- Location Olen eksyksissä. Not knowing where you are Voisitko näyttää kartalta missä sen on? Asking for a specific location on a map Mistä täällä on? Asking for a specific...wc?...pankki / rahanvaihtopiste?...hotelli?...huoltoasema?...sairaala?...apteekki?...tavaratalo?...ruokakauppa?...bussipysäkki?
OFFICE 365 OPISKELIJOILLE
 OFFICE 365 OPISKELIJOILLE Table of Contents Articles... 3 Ohjeet Office 365 käyttöönottoon... 4 One Driveen tallennetun videon palauttaminen oppimisympäristön palautuskansioon... 5 Changing default language
OFFICE 365 OPISKELIJOILLE Table of Contents Articles... 3 Ohjeet Office 365 käyttöönottoon... 4 One Driveen tallennetun videon palauttaminen oppimisympäristön palautuskansioon... 5 Changing default language
S Sähkön jakelu ja markkinat S Electricity Distribution and Markets
 S-18.3153 Sähkön jakelu ja markkinat S-18.3154 Electricity Distribution and Markets Voltage Sag 1) Kolmivaiheinen vastukseton oikosulku tapahtuu 20 kv lähdöllä etäisyydellä 1 km, 3 km, 5 km, 8 km, 10 km
S-18.3153 Sähkön jakelu ja markkinat S-18.3154 Electricity Distribution and Markets Voltage Sag 1) Kolmivaiheinen vastukseton oikosulku tapahtuu 20 kv lähdöllä etäisyydellä 1 km, 3 km, 5 km, 8 km, 10 km
HARJOITUS- PAKETTI A
 Logistiikka A35A00310 Tuotantotalouden perusteet HARJOITUS- PAKETTI A (6 pistettä) TUTA 19 Luento 3.Ennustaminen County General 1 piste The number of heart surgeries performed at County General Hospital
Logistiikka A35A00310 Tuotantotalouden perusteet HARJOITUS- PAKETTI A (6 pistettä) TUTA 19 Luento 3.Ennustaminen County General 1 piste The number of heart surgeries performed at County General Hospital
AYYE 9/ HOUSING POLICY
 AYYE 9/12 2.10.2012 HOUSING POLICY Mission for AYY Housing? What do we want to achieve by renting apartments? 1) How many apartments do we need? 2) What kind of apartments do we need? 3) To whom do we
AYYE 9/12 2.10.2012 HOUSING POLICY Mission for AYY Housing? What do we want to achieve by renting apartments? 1) How many apartments do we need? 2) What kind of apartments do we need? 3) To whom do we
SELL Student Games kansainvälinen opiskelijaurheilutapahtuma
 SELL Student Games kansainvälinen opiskelijaurheilutapahtuma Painonnosto 13.5.2016 (kansallinen, CUP) Below in English Paikka: Nääshalli Näsijärvenkatu 8 33210 Tampere Alustava aikataulu: Punnitus 12:00-13:00
SELL Student Games kansainvälinen opiskelijaurheilutapahtuma Painonnosto 13.5.2016 (kansallinen, CUP) Below in English Paikka: Nääshalli Näsijärvenkatu 8 33210 Tampere Alustava aikataulu: Punnitus 12:00-13:00
C++11 seminaari, kevät Johannes Koskinen
 C++11 seminaari, kevät 2012 Johannes Koskinen Sisältö Mikä onkaan ongelma? Standardidraftin luku 29: Atomiset tyypit Muistimalli Rinnakkaisuus On multicore systems, when a thread writes a value to memory,
C++11 seminaari, kevät 2012 Johannes Koskinen Sisältö Mikä onkaan ongelma? Standardidraftin luku 29: Atomiset tyypit Muistimalli Rinnakkaisuus On multicore systems, when a thread writes a value to memory,
Rakennukset Varjostus "real case" h/a 0,5 1,5
 Tuulivoimala Rakennukset Asuinrakennus Liikerak. tai Julkinen rak. Lomarakennus Teollinen rakennus Kirkollinen rakennus Varjostus "real case" h/a 1 h/a 8 h/a 20 h/a 1 2 3 5 8 4 6 7 9 10 0 0,5 1 1,5 2 km
Tuulivoimala Rakennukset Asuinrakennus Liikerak. tai Julkinen rak. Lomarakennus Teollinen rakennus Kirkollinen rakennus Varjostus "real case" h/a 1 h/a 8 h/a 20 h/a 1 2 3 5 8 4 6 7 9 10 0 0,5 1 1,5 2 km
Sisällysluettelo Table of contents
 Sisällysluettelo Table of contents OTC:n Moodlen käyttöohje suomeksi... 1 Kirjautuminen Moodleen... 2 Ensimmäinen kirjautuminen Moodleen... 2 Salasanan vaihto... 2 Oma käyttäjäprofiili... 3 Työskentely
Sisällysluettelo Table of contents OTC:n Moodlen käyttöohje suomeksi... 1 Kirjautuminen Moodleen... 2 Ensimmäinen kirjautuminen Moodleen... 2 Salasanan vaihto... 2 Oma käyttäjäprofiili... 3 Työskentely
812336A C++ -kielen perusteet, 21.8.2010
 812336A C++ -kielen perusteet, 21.8.2010 1. Vastaa lyhyesti seuraaviin kysymyksiin (1p kaikista): a) Mitä tarkoittaa funktion ylikuormittaminen (overloading)? b) Mitä tarkoittaa jäsenfunktion ylimääritys
812336A C++ -kielen perusteet, 21.8.2010 1. Vastaa lyhyesti seuraaviin kysymyksiin (1p kaikista): a) Mitä tarkoittaa funktion ylikuormittaminen (overloading)? b) Mitä tarkoittaa jäsenfunktion ylimääritys
Salasanan vaihto uuteen / How to change password
 Salasanan vaihto uuteen / How to change password Sisällys Salasanakäytäntö / Password policy... 2 Salasanan vaihto verkkosivulla / Change password on website... 3 Salasanan vaihto matkapuhelimella / Change
Salasanan vaihto uuteen / How to change password Sisällys Salasanakäytäntö / Password policy... 2 Salasanan vaihto verkkosivulla / Change password on website... 3 Salasanan vaihto matkapuhelimella / Change
Tutkimusdata ja julkaiseminen Suomen Akatemian ja EU:n H2020 projekteissa
 Tutkimusdata ja julkaiseminen Suomen Akatemian ja EU:n H2020 projekteissa Tutkimusasiamies Kaisa Männikkö Tutkimus- ja innovaatiopalvelut Suomen Akatemian projektit Suomen Akatemia kehottaa avoimeen tieteelliseen
Tutkimusdata ja julkaiseminen Suomen Akatemian ja EU:n H2020 projekteissa Tutkimusasiamies Kaisa Männikkö Tutkimus- ja innovaatiopalvelut Suomen Akatemian projektit Suomen Akatemia kehottaa avoimeen tieteelliseen
TIKIT a) Suorassa tikissä ristiommel jää nahan alle piiloon. b) Ristitikissä ommel jää näkyviin.
 CML WHEEL COVER -RUORINAHKAN OMPELUOHJE Liota nahkoja lämpimässä vedessä n. 15 minuuttia ennen ompelua. Pidä nahka kosteana koko ompelun ajan esim. sumutepullolla. Pidä ommellessa kevyt kireys nahkaan,
CML WHEEL COVER -RUORINAHKAN OMPELUOHJE Liota nahkoja lämpimässä vedessä n. 15 minuuttia ennen ompelua. Pidä nahka kosteana koko ompelun ajan esim. sumutepullolla. Pidä ommellessa kevyt kireys nahkaan,
Information on Finnish Language Courses Spring Semester 2018 Päivi Paukku & Jenni Laine Centre for Language and Communication Studies
 Information on Finnish Language Courses Spring Semester 2018 Päivi Paukku & Jenni Laine 4.1.2018 Centre for Language and Communication Studies Puhutko suomea? -Hei! -Hei hei! -Moi! -Moi moi! -Terve! -Terve
Information on Finnish Language Courses Spring Semester 2018 Päivi Paukku & Jenni Laine 4.1.2018 Centre for Language and Communication Studies Puhutko suomea? -Hei! -Hei hei! -Moi! -Moi moi! -Terve! -Terve
Information on Finnish Courses Autumn Semester 2017 Jenni Laine & Päivi Paukku Centre for Language and Communication Studies
 Information on Finnish Courses Autumn Semester 2017 Jenni Laine & Päivi Paukku 24.8.2017 Centre for Language and Communication Studies Puhutko suomea? -Hei! -Hei hei! -Moi! -Moi moi! -Terve! -Terve terve!
Information on Finnish Courses Autumn Semester 2017 Jenni Laine & Päivi Paukku 24.8.2017 Centre for Language and Communication Studies Puhutko suomea? -Hei! -Hei hei! -Moi! -Moi moi! -Terve! -Terve terve!
Kysymys 5 Compared to the workload, the number of credits awarded was (1 credits equals 27 working hours): (4)
 Tilasto T1106120-s2012palaute Kyselyn T1106120+T1106120-s2012palaute yhteenveto: vastauksia (4) Kysymys 1 Degree programme: (4) TIK: TIK 1 25% ************** INF: INF 0 0% EST: EST 0 0% TLT: TLT 0 0% BIO:
Tilasto T1106120-s2012palaute Kyselyn T1106120+T1106120-s2012palaute yhteenveto: vastauksia (4) Kysymys 1 Degree programme: (4) TIK: TIK 1 25% ************** INF: INF 0 0% EST: EST 0 0% TLT: TLT 0 0% BIO:
Information on Finnish Language Courses Spring Semester 2017 Jenni Laine
 Information on Finnish Language Courses Spring Semester 2017 Jenni Laine 4.1.2017 KIELIKESKUS LANGUAGE CENTRE Puhutko suomea? Do you speak Finnish? -Hei! -Moi! -Mitä kuuluu? -Kiitos, hyvää. -Entä sinulle?
Information on Finnish Language Courses Spring Semester 2017 Jenni Laine 4.1.2017 KIELIKESKUS LANGUAGE CENTRE Puhutko suomea? Do you speak Finnish? -Hei! -Moi! -Mitä kuuluu? -Kiitos, hyvää. -Entä sinulle?
ECVETin soveltuvuus suomalaisiin tutkinnon perusteisiin. Case:Yrittäjyyskurssi matkailualan opiskelijoille englantilaisen opettajan toteuttamana
 ECVETin soveltuvuus suomalaisiin tutkinnon perusteisiin Case:Yrittäjyyskurssi matkailualan opiskelijoille englantilaisen opettajan toteuttamana Taustaa KAO mukana FINECVET-hankeessa, jossa pilotoimme ECVETiä
ECVETin soveltuvuus suomalaisiin tutkinnon perusteisiin Case:Yrittäjyyskurssi matkailualan opiskelijoille englantilaisen opettajan toteuttamana Taustaa KAO mukana FINECVET-hankeessa, jossa pilotoimme ECVETiä
ENE-C2001 Käytännön energiatekniikkaa. Aloitustapaaminen 11.4.2016. Osa II: Projekti- ja tiimityö
 ENE-C2001 Käytännön energiatekniikkaa Aloitustapaaminen 11.4.2016 Osa II: Projekti- ja tiimityö Sisältö Projektityö Mitä on projektityö? Projektityön tekeminen: ositus, aikatauluhallinta, päätöksenteon
ENE-C2001 Käytännön energiatekniikkaa Aloitustapaaminen 11.4.2016 Osa II: Projekti- ja tiimityö Sisältö Projektityö Mitä on projektityö? Projektityön tekeminen: ositus, aikatauluhallinta, päätöksenteon
1.3Lohkorakenne muodostetaan käyttämällä a) puolipistettä b) aaltosulkeita c) BEGIN ja END lausekkeita d) sisennystä
 OULUN YLIOPISTO Tietojenkäsittelytieteiden laitos Johdatus ohjelmointiin 81122P (4 ov.) 30.5.2005 Ohjelmointikieli on Java. Tentissä saa olla materiaali mukana. Tenttitulokset julkaistaan aikaisintaan
OULUN YLIOPISTO Tietojenkäsittelytieteiden laitos Johdatus ohjelmointiin 81122P (4 ov.) 30.5.2005 Ohjelmointikieli on Java. Tentissä saa olla materiaali mukana. Tenttitulokset julkaistaan aikaisintaan
FinFamily Installation and importing data (11.1.2016) FinFamily Asennus / Installation
 FinFamily Asennus / Installation 1 Sisällys / Contents FinFamily Asennus / Installation... 1 1. Asennus ja tietojen tuonti / Installation and importing data... 4 1.1. Asenna Java / Install Java... 4 1.2.
FinFamily Asennus / Installation 1 Sisällys / Contents FinFamily Asennus / Installation... 1 1. Asennus ja tietojen tuonti / Installation and importing data... 4 1.1. Asenna Java / Install Java... 4 1.2.
ALOITUSKESKUSTELU / FIRST CONVERSATION
 ALOITUSKESKUSTELU / FIRST CONVERSATION Lapsen nimi / Name of the child Lapsen ikä / Age of the child yrs months HYVINKÄÄN KAUPUNKI Varhaiskasvatuspalvelut Lapsen päivähoito daycare center / esiopetusyksikkö
ALOITUSKESKUSTELU / FIRST CONVERSATION Lapsen nimi / Name of the child Lapsen ikä / Age of the child yrs months HYVINKÄÄN KAUPUNKI Varhaiskasvatuspalvelut Lapsen päivähoito daycare center / esiopetusyksikkö
Rekisteröiminen - FAQ
 Rekisteröiminen - FAQ Miten Akun/laturin rekisteröiminen tehdään Akun/laturin rekisteröiminen tapahtuu samalla tavalla kuin nykyinen takuurekisteröityminen koneille. Nykyistä tietokantaa on muokattu niin,
Rekisteröiminen - FAQ Miten Akun/laturin rekisteröiminen tehdään Akun/laturin rekisteröiminen tapahtuu samalla tavalla kuin nykyinen takuurekisteröityminen koneille. Nykyistä tietokantaa on muokattu niin,
Lab A1.FARM_Hyper-V.v3
 Lab A1.FARM_Hyper-V Installing SharePoint Server 2013 SharePoint Server 2013 -asennus Scenario To install and configure SharePoint 2013 on a single server (Server 2012, AD and SQL Server), you will follow
Lab A1.FARM_Hyper-V Installing SharePoint Server 2013 SharePoint Server 2013 -asennus Scenario To install and configure SharePoint 2013 on a single server (Server 2012, AD and SQL Server), you will follow
Microsoft Lync 2010 Attendee
 VYVI MEETING Lync Attendee 2010 Instruction 1 (15) Microsoft Lync 2010 Attendee Online meeting VYVI MEETING Lync Attendee 2010 Instruction 2 (15) Index 1 Microsoft LYNC 2010 Attendee... 3 2 Acquiring Lync
VYVI MEETING Lync Attendee 2010 Instruction 1 (15) Microsoft Lync 2010 Attendee Online meeting VYVI MEETING Lync Attendee 2010 Instruction 2 (15) Index 1 Microsoft LYNC 2010 Attendee... 3 2 Acquiring Lync
Oma sininen meresi (Finnish Edition)
 Oma sininen meresi (Finnish Edition) Hannu Pirilä Click here if your download doesn"t start automatically Oma sininen meresi (Finnish Edition) Hannu Pirilä Oma sininen meresi (Finnish Edition) Hannu Pirilä
Oma sininen meresi (Finnish Edition) Hannu Pirilä Click here if your download doesn"t start automatically Oma sininen meresi (Finnish Edition) Hannu Pirilä Oma sininen meresi (Finnish Edition) Hannu Pirilä
Guidebook for Multicultural TUT Users
 1 Guidebook for Multicultural TUT Users WORKPLACE PIRKANMAA-hankkeen KESKUSTELUTILAISUUS 16.12.2010 Hyvää käytäntöä kehittämässä - vuorovaikutusopas kansainvälisille opiskelijoille TTY Teknis-taloudellinen
1 Guidebook for Multicultural TUT Users WORKPLACE PIRKANMAA-hankkeen KESKUSTELUTILAISUUS 16.12.2010 Hyvää käytäntöä kehittämässä - vuorovaikutusopas kansainvälisille opiskelijoille TTY Teknis-taloudellinen
Uusia kokeellisia töitä opiskelijoiden tutkimustaitojen kehittämiseen
 The acquisition of science competencies using ICT real time experiments COMBLAB Uusia kokeellisia töitä opiskelijoiden tutkimustaitojen kehittämiseen Project N. 517587-LLP-2011-ES-COMENIUS-CMP This project
The acquisition of science competencies using ICT real time experiments COMBLAB Uusia kokeellisia töitä opiskelijoiden tutkimustaitojen kehittämiseen Project N. 517587-LLP-2011-ES-COMENIUS-CMP This project
Kaivostoiminnan eri vaiheiden kumulatiivisten vaikutusten huomioimisen kehittäminen suomalaisessa luonnonsuojelulainsäädännössä
 M a t t i K a t t a i n e n O T M 1 1. 0 9. 2 0 1 9 Kaivostoiminnan eri vaiheiden kumulatiivisten vaikutusten huomioimisen kehittäminen suomalaisessa luonnonsuojelulainsäädännössä Ympäristöoikeustieteen
M a t t i K a t t a i n e n O T M 1 1. 0 9. 2 0 1 9 Kaivostoiminnan eri vaiheiden kumulatiivisten vaikutusten huomioimisen kehittäminen suomalaisessa luonnonsuojelulainsäädännössä Ympäristöoikeustieteen
Business Opening. Arvoisa Herra Presidentti Very formal, recipient has a special title that must be used in place of their name
 - Opening Finnish Norwegian Arvoisa Herra Presidentti Very formal, recipient has a special title that must be used in place of their name Hyvä Herra, Formal, male recipient, name unknown Hyvä Rouva Formal,
- Opening Finnish Norwegian Arvoisa Herra Presidentti Very formal, recipient has a special title that must be used in place of their name Hyvä Herra, Formal, male recipient, name unknown Hyvä Rouva Formal,
Exercise 1. (session: )
 EEN-E3001, FUNDAMENTALS IN INDUSTRIAL ENERGY ENGINEERING Exercise 1 (session: 24.1.2017) Problem 3 will be graded. The deadline for the return is on 31.1. at 12:00 am (before the exercise session). You
EEN-E3001, FUNDAMENTALS IN INDUSTRIAL ENERGY ENGINEERING Exercise 1 (session: 24.1.2017) Problem 3 will be graded. The deadline for the return is on 31.1. at 12:00 am (before the exercise session). You
EVALUATION FOR THE ERASMUS+-PROJECT, STUDENTSE
 #1 Aloitettu: 6. marraskuuta 2015 9:03:38 Muokattu viimeksi: 6. marraskuuta 2015 9:05:26 Käytetty aika: 00:01:47 IP-osoite: 83.245.241.86 K1: Nationality Finnish K2: The program of the week has been very
#1 Aloitettu: 6. marraskuuta 2015 9:03:38 Muokattu viimeksi: 6. marraskuuta 2015 9:05:26 Käytetty aika: 00:01:47 IP-osoite: 83.245.241.86 K1: Nationality Finnish K2: The program of the week has been very
Green Growth Sessio - Millaisilla kansainvälistymismalleilla kasvumarkkinoille?
 Green Growth Sessio - Millaisilla kansainvälistymismalleilla kasvumarkkinoille? 10.10.01 Tuomo Suortti Ohjelman päällikkö Riina Antikainen Ohjelman koordinaattori 10/11/01 Tilaisuuden teema Kansainvälistymiseen
Green Growth Sessio - Millaisilla kansainvälistymismalleilla kasvumarkkinoille? 10.10.01 Tuomo Suortti Ohjelman päällikkö Riina Antikainen Ohjelman koordinaattori 10/11/01 Tilaisuuden teema Kansainvälistymiseen
EUROOPAN PARLAMENTTI
 EUROOPAN PARLAMENTTI 2004 2009 Kansalaisvapauksien sekä oikeus- ja sisäasioiden valiokunta 2008/0101(CNS) 2.9.2008 TARKISTUKSET 9-12 Mietintöluonnos Luca Romagnoli (PE409.790v01-00) ehdotuksesta neuvoston
EUROOPAN PARLAMENTTI 2004 2009 Kansalaisvapauksien sekä oikeus- ja sisäasioiden valiokunta 2008/0101(CNS) 2.9.2008 TARKISTUKSET 9-12 Mietintöluonnos Luca Romagnoli (PE409.790v01-00) ehdotuksesta neuvoston
TIETEEN PÄIVÄT OULUSSA 1.-2.9.2015
 1 TIETEEN PÄIVÄT OULUSSA 1.-2.9.2015 Oulun Yliopisto / Tieteen päivät 2015 2 TIETEEN PÄIVÄT Järjestetään Oulussa osana yliopiston avajaisviikon ohjelmaa Tieteen päivät järjestetään saman konseptin mukaisesti
1 TIETEEN PÄIVÄT OULUSSA 1.-2.9.2015 Oulun Yliopisto / Tieteen päivät 2015 2 TIETEEN PÄIVÄT Järjestetään Oulussa osana yliopiston avajaisviikon ohjelmaa Tieteen päivät järjestetään saman konseptin mukaisesti
Group 2 - Dentego PTH Korvake. Peer Testing Report
 Group 2 - Dentego PTH Korvake Peer Testing Report Revisions Version Date Author Description 1.0 Henrik Klinkmann First version Table of Contents Contents Revisions... 2 Table of Contents... 2 Testing...
Group 2 - Dentego PTH Korvake Peer Testing Report Revisions Version Date Author Description 1.0 Henrik Klinkmann First version Table of Contents Contents Revisions... 2 Table of Contents... 2 Testing...
I. Principles of Pointer Year Analysis
 I. Principles of Pointer Year Analysis Fig 1. Maximum (red) and minimum (blue) pointer years. 1 Fig 2. Principle of pointer year calculation. Fig 3. Skeleton plot graph created by Kinsys/Kigraph programme.
I. Principles of Pointer Year Analysis Fig 1. Maximum (red) and minimum (blue) pointer years. 1 Fig 2. Principle of pointer year calculation. Fig 3. Skeleton plot graph created by Kinsys/Kigraph programme.
Arkkitehtuuritietoisku. eli mitä aina olet halunnut tietää arkkitehtuureista, muttet ole uskaltanut kysyä
 Arkkitehtuuritietoisku eli mitä aina olet halunnut tietää arkkitehtuureista, muttet ole uskaltanut kysyä Esikysymys Kuinka moni aikoo suunnitella projektityönsä arkkitehtuurin? Onko tämä arkkitehtuuria?
Arkkitehtuuritietoisku eli mitä aina olet halunnut tietää arkkitehtuureista, muttet ole uskaltanut kysyä Esikysymys Kuinka moni aikoo suunnitella projektityönsä arkkitehtuurin? Onko tämä arkkitehtuuria?
Rotarypiiri 1420 Piiriapurahoista myönnettävät stipendit
 Rotarypiiri 1420 Piiriapurahoista myönnettävät stipendit Ø Rotarypiiri myöntää stipendejä sille osoitettujen hakemusten perusteella ensisijaisesti rotaryaatteen mukaisiin tarkoituksiin. Ø Stipendejä myönnetään
Rotarypiiri 1420 Piiriapurahoista myönnettävät stipendit Ø Rotarypiiri myöntää stipendejä sille osoitettujen hakemusten perusteella ensisijaisesti rotaryaatteen mukaisiin tarkoituksiin. Ø Stipendejä myönnetään
VUOSI 2015 / YEAR 2015
 VUOSI 2015 / YEAR 2015 Kansainvälisen opetuksen ja tutkimustoiminnan kehittäminen Developing international teaching and research activities Rehtorin strateginen rahoitus vuosille 2014-2016 / Strategic
VUOSI 2015 / YEAR 2015 Kansainvälisen opetuksen ja tutkimustoiminnan kehittäminen Developing international teaching and research activities Rehtorin strateginen rahoitus vuosille 2014-2016 / Strategic
